The recent buzz surrounding the FDA's decision on BPC 157, a peptide renowned for its potential healing properties, has created quite a stir. This decision has led to varied opinions and discussions in the health community. In this article, we delve into the diverse perspectives on BPC 157's benefits and the FDA's decision, with a particular focus on why Dr. Rocco Piazza remains committed to offering BPC 157 in oral form.
Benefits of BPC-157 and the FDA's Concerns
BPC-157, a peptide derived from a protein in the stomach, has been studied for its potential health benefits. Despite headlines suggesting a ban, the FDA has simply placed BPC-157 in a category requiring further investigation. This nuanced position calls for a deeper understanding of BPC-157's origins and therapeutic potential amidst evolving regulatory perspectives.
Reported Benefits of BPC-157:
- Accelerated Healing: Studies suggest that BPC-157 may accelerate healing of wounds, muscles, tendons, and nerves.
- Inflammation Reduction: It is reported to have anti-inflammatory effects, potentially offering relief for various conditions.
- Gastrointestinal Health: Research indicates BPC-157 could improve digestive disorders, such as healing ulcers and mitigating NSAID-induced damage.
- Musculoskeletal Repair: Evidence suggests it may aid in repairing damaged ligaments and tendons.
- Cardiovascular and Mental Health: Preliminary research hints at benefits for cardiovascular health and mental conditions like depression and anxiety.
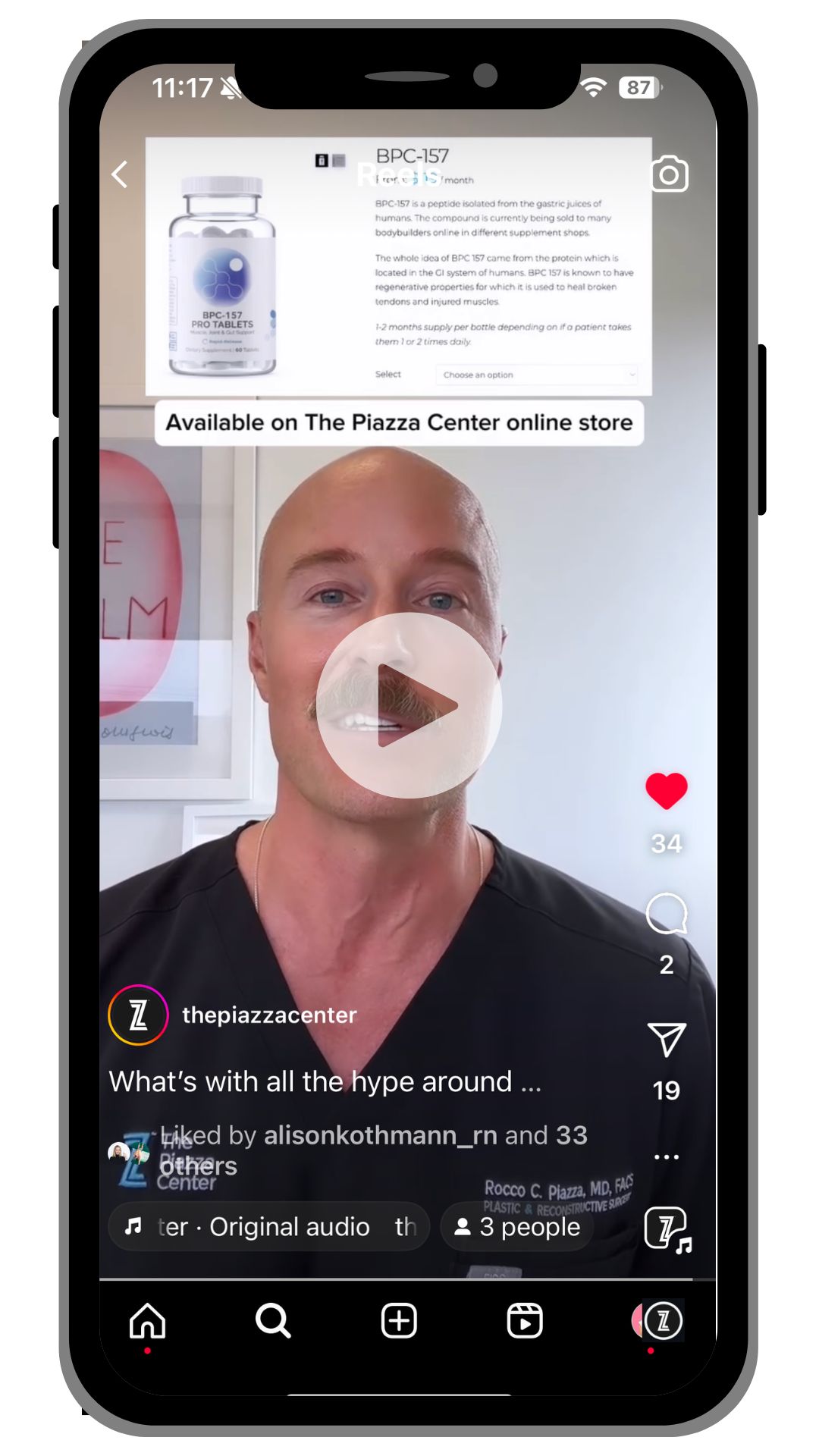
Why The Piazza Center still offers BPC 157 oral supplement
Amidst this controversy, Dr. Rocco Piazza stands firm in his decision to continue offering BPC 157 in oral form. He believes that this supplement, which remains under different regulations as a supplement in oral form, holds significant promise for patient care. As always, before starting any drug or supplement, if you have concerns, please ensure you speak to your general practitioner to ensure you are medically cleared to take this supplement for gut health.
While BPC-157 injections face restrictions due to regulatory, safety, and efficacy concerns, the pill form can still be sold as a supplement under different, less stringent regulations. However, consumers should exercise caution and consult healthcare providers before using such supplements, as their safety and efficacy might not be well-established. It is Dr. Piazza’s belief that the benefits of using BPC-157 as an oral supplement outweigh the concerns as outline by the FDA for the injection of BPC-157.
Watch more:
Reasons for Dr. Piazza’s Decision:
- Patient Demand and Reported Benefits: Patients have reported positive outcomes using oral BPC 157, particularly in areas like accelerated healing and inflammation reduction.
- Holistic Health Approach: Dr. Piazza advocates for a holistic health approach, offering alternatives that may benefit those not fully addressed by traditional treatments.
- Commitment to Innovation: Believing in the potential of new health solutions, Dr. Piazza aims to provide access to innovative therapies while prioritizing patient safety.
Despite these promising benefits, the FDA's decision to scrutinize BPC-157 is based on several key factors.
The FDA's ban on BPC-157 peptide
The FDA's concerns about BPC 157 center on safety considerations and the absence of comprehensive clinical trials. The FDA's classification indicates the need for more rigorous investigation, impacting the availability and distribution of BPC 157.
FDA's Official Reasons:
- Lack of Clinical Trials: The FDA emphasizes the need for extensive, controlled clinical trials, which are currently lacking.
- Safety Concerns: Without comprehensive human trials, the FDA cannot assure the safety of BPC-157, including potential side effects, long-term impacts, and appropriate dosages.
- Regulatory Compliance: The FDA highlights the importance of regulatory compliance, which BPC-157 has not fully met according to their standards.
Consequences of FDA’s Classification
The FDA's decision has led to changes in BPC-157's availability, sparking debates within the medical and wellness communities about regulatory processes and the challenges in bringing new therapies to market.
Controversy and Diverse Perspectives on BPC-157
While BPC 157 is not officially banned, the FDA's classification has ignited discussions among health experts and supporters of alternative therapies. Critics argue that the FDA's caution could restrict access to a potentially beneficial compound, advocating for a more balanced approach that considers BPC-157's therapeutic potential.
Arguments Against the FDA’s Decision:
- Potential Health Benefits: Advocates emphasize the reported benefits observed in animal studies and limited human use, arguing these should be weighed alongside regulatory concerns.
- Need for Progressive Regulation: Critics suggest the FDA’s regulatory processes may not fully accommodate the fast-paced evolution of biomedical research.
- Global Perspective: BPC-157's use in other countries with reported success raises questions about the FDA's stricter stance compared to other regions.
- Big Pharma’s Role: Speculation about pharmaceutical companies influencing the FDA’s decision highlights concerns about competition and access to new treatments.
Conclusion
Navigating the evolving landscape of health and wellness treatments requires staying informed and advocating for balanced healthcare solutions. Despite the FDA's scrutiny, Dr. Rocco Piazza's commitment to offering BPC 157 in oral form underscores the ongoing debate between regulatory caution and access to promising health treatments. At The Piazza Center, we support informed and innovative care. For more about our alternative therapies and personalized support, visit our services page or contact us to learn how we can help you achieve optimal health and wellness.
For more information, please visit our online store. For prescription BPC, we also recommend working with the clinica team at Celia Health.
FDA Disclaimer
- General Disclaimer: These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- Usage and Safety Disclaimers
- Consultation with Healthcare Provider: Consult your healthcare provider before using this product if you are pregnant, nursing, taking any medications, or have any medical conditions.
- Not for Specific Populations: Not intended for use by individuals under the age of 18.
- Keep Out of Reach of Children:
- Additional Disclaimers
- Intended Use: “This product is intended for dietary supplement use only and should not be used for any other purposes.”
- Possible Side Effects: “Discontinue use and consult your healthcare provider if any adverse reactions occur.”